IZiel Healthcare is built on the foundation of technical excellence, sound management practices and winning customer trust. IZiel Healthcare provides Engineering & Regulatory Services for global Class I, II & III medical device companies from our offices in USA & India.
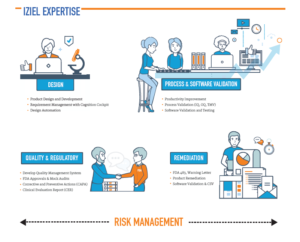
IZiel Engineers are trained in Design for Six Sigma (DFSS) and have completed projects in –
- Design Documentation
- Process Validation (IQ, OQ, PQ, TMV)
- Software Validation, Testing & Cybersecurity
- Tool (CSV) Validation
- Risk Management
- Regulatory Approvals (USFDA, CE)
- Acquisition Integration
- Remediation
Our Outcome-Based & Cost-Effective Model using global teams and comprehensive monitoring system have ensured effective and timely deliveries for medical device companies.
Team Members

Asavari Dandekar
Partner

Durga Kulkarni
Partner

Ankur Naik
Partner